Spanish drug packaging company Farma-Química Sur reportedly mislabeled different drugs, resulting in the release of medicine to children which has caused abnormal hair growth and poses a deadly risk of premature death to babies. The company nevertheless holds ISO 9001 and ISO 14001 certifications issued by TUV Rheinland Iberica, accredited by the Spanish accreditation body ENAC.
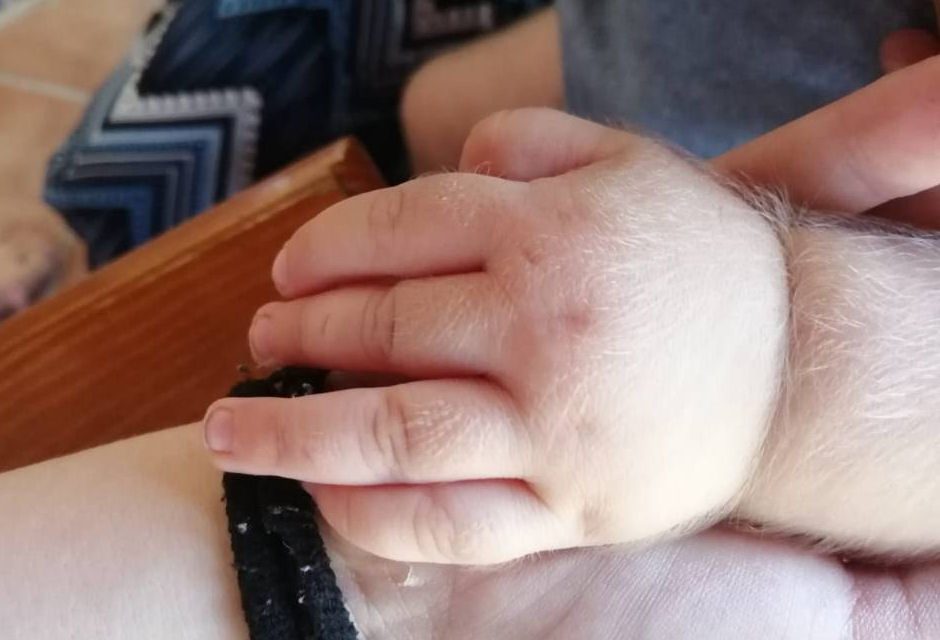
Farma-Química Sur allegedly mislabeled gastritis medicine omeprazole as Minoxidil, a common medicine used to treat baldness. This resulted in at least 20 babies, some younger than 2 years old, suffering from uncontrolled all-over hair growth and other symptoms. According to reporting from the Associated Press, “Spain’s drug agency, AEMPS, says Minoxidil’s ability to widen blood vessels could impact the children’s hearts, kidneys and livers in the future.”
 The ISO 9001 certificate issued to Farma-Química Sur by TUV was granted in June of 2019 and is valid until 2022. So far, TUV has not withdrawn the certificate, despite widespread regional reporting of the mishap. Representatives of TUV Rheinland Iberica did not immediately reply to requests for information by Oxebridge.
The ISO 9001 certificate issued to Farma-Química Sur by TUV was granted in June of 2019 and is valid until 2022. So far, TUV has not withdrawn the certificate, despite widespread regional reporting of the mishap. Representatives of TUV Rheinland Iberica did not immediately reply to requests for information by Oxebridge.
ISO 9001 audits by TUV should have confirmed Farma-Química Sur’s processes for product identification as part of assessing its compliance to clause 8.5.2.
TUV Rheinland Iberica is accredited by ENAC, which itself is a member of the IAF. To date, despite over a hundred reports of ISO 9001 certificates being issued by IAF-matrixed registrars to companies involved in deadly product releases, scandals, crimes or disasters, the IAF has not taken action against a single party involved. Some 40 certification bodies, and 12 accreditation bodies, are involved. All are traced back to IAF membership by the applicable accreditation body.
Of the reports being monitored by Oxebridge, over 1,700 people have died at the hands of companies holding ISO certificates bearing the IAF logo.
UPDATE 5 Sept 2019: TUV has responded to this report on Twitter, tweeting: [Photo courtesy El Pais.]
[Photo courtesy El Pais.]